Field defect, high IOP might not signal glaucoma
Glaucoma is a term that describes a family of progressive optic neuropathies. All of the glaucomas share characteristic and progressive cupping of the optic nerve head, and this cupping is most easily viewed by means of direct stereoscopic evaluation through a dilated pupil.
Large optic nerve heads may have physiologically large optic cups, while smaller optic nerve heads tend to have smaller optic cups. A large optic nerve head with a large cup may very well have about the same number of ganglion cells traversing through it as a smaller optic nerve head with a smaller cup. Some optic nerve heads are so small that they are classified as being congenitally hypoplastic (congenital optic nerve hypoplasia [CONH]). With respect to these optic nerves, it is often difficult to ascertain the presence of an optic cup at all.
Patients with CONH may very well go on to develop other acquired diseases of their optic nerves unrelated to their CONH. Moreover, since visual field defects are not uncommon in patients with CONH,1 it may be difficult to determine if a newly discovered visual field defect is related to the hypoplasticity of the optic disc (relatively static) or some other cause such as glaucoma (relatively progressive), especially if the visual field defect is nasal in location.
Case presentation
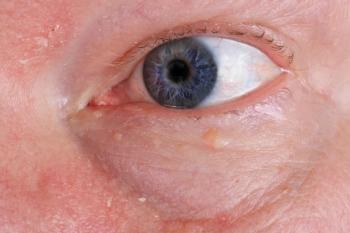
With this in mind, I was asked to examine a patient not long ago to help in confirming or denying the presence of glaucoma. The patient was a 68-year-old white female with a medical history remarkable for arterial hypertension, hypercholesterolemia, and hypothyroidism-all reportedly under good control. Medications included hydrochlorothiazide (Microzide, Actavis), simvastatin (Zocor, Merck), and levothyroxine (Tirosint, Akrimax). The patient had no other complaints or apparent ocular concerns. Best-corrected visual acuities were 20/25 OU, and slit lamp examination was remarkable for moderate nuclear cataracts in each eye. Pupils were unremarkable in each eye. Intraocular pressures (IOP) by means of Goldmann applanation tonometry were 26 mm Hg OD and 28 mm Hg OS at 1:45 p.m. Photos of the patient’s optic nerve heads after pupillary dilation are as shown in Figure 1. Peripheral retina evaluation via indirect ophthalmoscopy was unremarkable, as was the remainder of the examination.
Right away, the patient’s optic nerve heads appeared to be very small. In fact, the right optic nerve head measured just 1.1 mm vertically, while the left optic nerve head measured 1.15 mm (average optic nerve head sizes vary by study and race, but they are commonly described as being 1.5 to 2 times greater than those of this patient).2 The vasculature also emanated from each optic nerve head in a somewhat radial fashion, another potential indicator for some sort of congenital malformation. It was difficult to determine the presence of any discernable cup, and there was also a faint scleral canal visible around each optic nerve head. As well, the retinal nerve fiber layer (RNFL) in each eye was noticeably thin virtually 360 degrees around each optic nerve head.
After taking fundus photos of each eye, I reminded the patient that a glaucoma work-up was a stepwise process and invited her back the following week (some time in the morning) for some glaucoma-specific testing. At that visit, visual acuities were unchanged. IOPs were 27 mm Hg OD and 28 mm Hg OS at 10:15 a.m. Pachymetry values were 533 µm OD and 536 µm OS. Gonioscopy showed that both eyes had angles open to the ciliary body with a flat iris approach and trace trabecular meshwork pigment in all four quadrants. Optical coherence tomography (OCT) studies confirmed the high degree of RNFL thinning seen upon fundoscopy (see Figure 2). The 24-2 threshold visual field studies showed significant nasal visual field defects in each eye (see Figure 3).
The gorillas in the room
Upon reviewing all of this exam data, it becomes apparent that the patient in question exhibits several diagnostic signs that happen to be characteristic of glaucoma, including RNFL thinning and nasal visual field defects. Both of these findings are commonly seen with CONH, as well. The 800-pound gorilla in the room (for me at least) was the presence of ocular hypertension (the number-one risk factor for the development of glaucoma). The other gorilla in the room, if you will, was fact that I could not discern an optic cup, per se, in either eye. The OCT studies helped me to visualize a very small cup displaced temporally in each eye, but glaucoma involves cupping of the optic nerve heads. So, I was uncomfortable with a diagnosis of glaucoma. One thing I was quite comfortable with, however, was the fact that any degree of noticeable cupping with respect to such small nerves would likely not be readily visible until a substantial number of ganglion cells had atrophied. This would present the scenario of a very small optic nerve head with a small, yet glaucomatous cup.
I told the patient that I frankly didn’t know whether or not she had glaucoma but doubted it. However, given her visual field defects, be they glaucomatous or not, her high eye pressures were presenting a risk for progression of these defects, and a modifiable risk at that. We had a brief discussion regarding the nature of glaucoma, and I decided to label her as a high-risk glaucoma suspect and treat her IOPs with a prostaglandin analog. I will recheck her IOPs and perform different visual field studies in a few weeks, and, although, she is older, I’ve decided it’s not a bad idea to order an MRI. (I’m not necessarily looking for tumors or bleeds but rather looking for other neurological dysplasias potentially correlated with her optic nerve hypoplasia).
Lurking variables are frequently present when working on a glaucoma diagnosis and have the capacity to complicate matters. If this patient’s intraocular pressures were not high, I would have likely watched her over a period of months for change by means of trend and event analysis using OCT and visual field studies. However, given her findings, I felt better about doing at least something to lessen her risk of visual problems down the road.
References
1. Kim US, Baek SH, Lee JH. Characteristics of segmental optic nerve hypoplasia. Eye (Lond). 2012 Dec;26(12):1585-6.
2. Jonas JB, Gusek GC, Naumann GO. Optic disc, cup, and neuroretinal rim size, configuration and correlation in normal eyes. Invest Ophthalmol Vis Sci. 1988 Jul;29(27):1151-8.
Newsletter
Want more insights like this? Subscribe to Optometry Times and get clinical pearls and practice tips delivered straight to your inbox.