Dry age-related macular degeneration: The second city of AMD
Prevention should be at the forefront of the discussion.
Age-related macular degeneration (AMD) represents a considerable burden with many facets. Considering the epidemiological, public health, fiscal, and individual impacts, AMD may be the most devastating ophthalmic disease. Despite recent developments for management of both wet (neovascular) and dry (non-neovascular) AMD, as well as geographic atrophy (GA), vision preservation remains the holy grail for practitioners and patients.
AMD is more than twice as prevalent as Alzheimer disease, and outstrips the prevalence of both diabetic retinopathy (DR) and glaucoma.1-3 Currently, AMD affects at least 20 million adults (> 40 years of age) in the United States.4 This includes those living with both vision-threatening and non–vision-threatening types of the condition, but does not include those affected as caregivers or employers, for example, according to the CDC.4 Prevalence increases exponentially with increasing age, especially over 80 years. According to projections from the National Eye Institute,5 the problem is not expected to abate.
Every February is dedicated to AMD awareness in a campaign by Prevent Blindness.6 This effort aims to reduce vision loss due to AMD, as it is well documented that over 75% of patients receiving treatment have already suffered irreversible vision loss.7 Although this statistic may be somewhat dated, the fact remains that outcomes for patients who receive an AMD diagnosis are guarded at best. Data from recent large-scale reports suggest there are minimal visual performance increases among older patients, even with conscientious adherence to treatment protocols.8,9 This indicates that just as with other chronic diseases and disorders, early detection and intervention may be beneficial for optimum outcomes.
Recent data suggest that ophthalmic clinicians are accurate in only about three-fourths of instances for detecting early AMD degeneration, even knowing that the patients were being recruited for an AMD study. The Alabama Study on Early Age-Related Macular Degeneration (ALSTAR) results showed that 28% of patients diagnosed as having early AMD by trained readers reviewing color fundus photographs were overlooked at clinical examination.10 The light at the end of this tunnel is that most cases showed only early fundus changes; but the finding also underscores the fact that as clinicians we could do a better job of adhering to the Beckman staging scale developed from the Age-Related Eye Disease Studies (AREDS).11 A highlight from this classification is that the presence of even small drusen (less than 60 µm, or half a retinal vein width) qualify as early AMD. More sophisticated means to detect and stage retinal changes in aging are on the horizon and will be aided by artificial intelligence.12
When I began my optometric journey in the mid-1970s, patients who developed AMD had little hope for vision stabilization, let alone improvement. In those days the terminology was “senile macular degeneration,” abbreviated SMD. William Feinbloom, OD, often acknowledged as the father of low-vision care, was a clinical faculty member at Pennsylvania College of Optometry, now Pennsylvania College of Optometry at Salus University in Elkins Park. He established the Feinbloom Low Vision Center.

Who reading this column has not used a Feinbloom chart? Patients were referred to Feinbloom’s clinic for vision rehabilitation in the hope of providing some measure of reading vision. Vision enhancement for driving was rarely a consideration for advanced cases. These patients were colloquially referred to as “Feinbloom patients” (Figure 1). An example of dry AMD is represented by drusen and significant GA in the Figure. Figure 2 shows a patient with significant drusen presentation and pigmentary abnormalities whose fellow eye had developed the neovascular form of AMD.
Although AMD is a significant problem from several aspects, the most important impact is on the patient. In a study, Gary Brown, MD, measured visual performance related to quality of life among patients with AMD, with findings published in Transactions of the American Ophthalmological Society journal.13 The study results showed that patients limited to just 20/40 visual acuity would trade 2 of their remaining 10 years of life for perfect vision and that patients with severe vision impairment would be willing to trade half their remaining 10 years for perfect vision.
The above data have directed much of the research into treatment for neovascular AMD. Laser photocoagulation, which ablated newly growing vessels with the aim of stabilizing the progression of proliferation, was the beginning of this research. This strategy failed in many cases due to collateral damage that allowed additional neovascularization.14,15 Because of these and other limitations, new and better approaches to the management of neovascular AMD were underway.
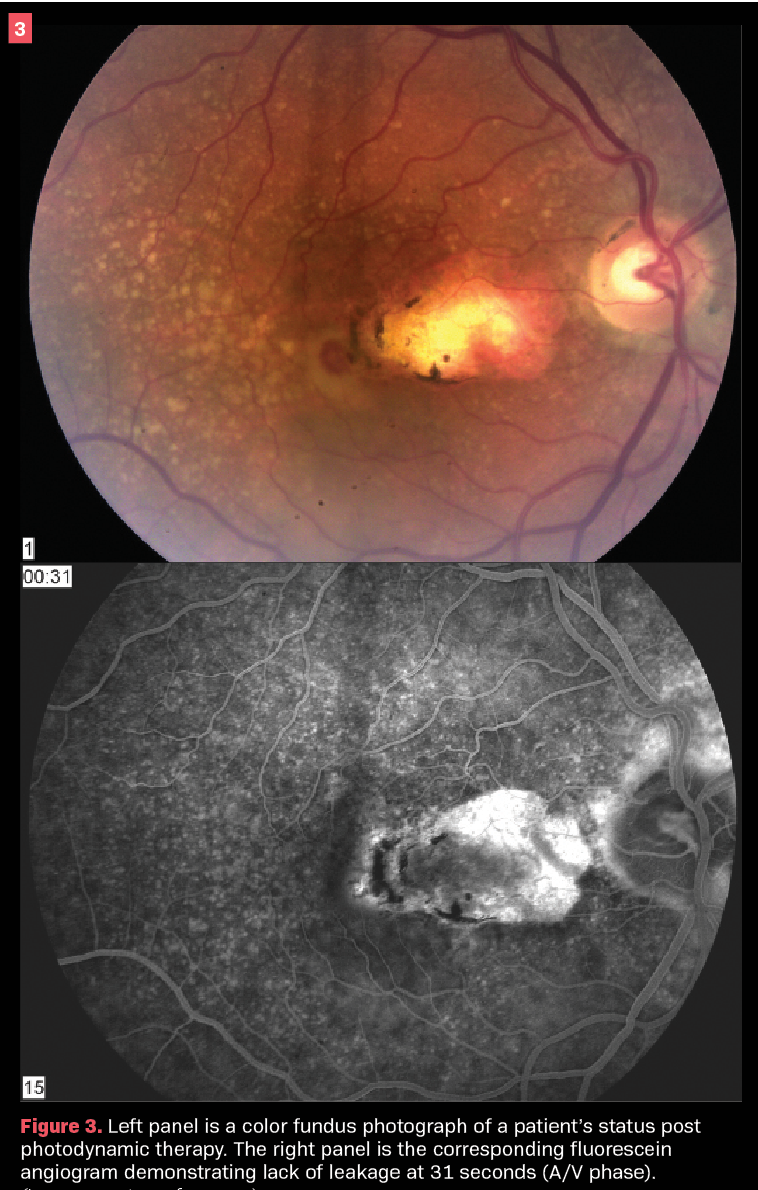
The first commercially available treatment was photodynamic therapy (PDT). The objective was to limit, but not eliminate, the neovascular process so that a nutritional supply could continue to serve the other retina.16 The protocol for PDT involved injection of the drug into the ante-cubital vein and subsequent irradiation (activation) at the slit lamp. Visual acuity stabilization was the goal.
In Figure 3, we see a 62-year-old patient with 20/200 visual acuity and accompanying full-thickness macular hole. Interestingly, the patient later underwent intravitreal ranibizumab injection as further treatment.
Images courtesty of Dr Leo Semes
Stabilization of visual acuity, which was state of the science over 2 decades ago, along with the significance of visual performance, ushered in the anti-angiogenic era. A pioneer was Judah Folkman, MD, whose work with angiogenesis inhibition for cancer was extrapolated to the eye.17,18 The first of these compounds was pegaptanib (Macugen) for neovascular AMD in 2004.19 It was demonstrated to be safe and effective for the treatment of neovascular AMD. At the molecular level, pegaptanib efficacy was limited to targeting a single isomer in the angiogenesis cascade. The application was quickly extended to other ophthalmic macular disorders.20 For AMD, the outcome still was limited to slowing the progression of the disease and limiting vision loss.21 The criterion was the number of patients losing fewer than 15 letters (Early Treatment Diabetic Retinopathy Study chart). Clearly there was room for improvement.
One such promising application, bevacizumab (Avastin), was not approved by the FDA for use in the eye. Intravitreal injection was pioneered by Philip J. Rosenfeld, MD, PhD, and it represented a revolution in the treatment of neovascular AMD.22 At about the same time, FDA approval was granted to a cousin of bevacizumab, ranibizumab (Lucentis). Controversy over the cost, efficacy, adverse effects, and outcomes between the 2 medications resulted in the CATT study, which demonstrated essentially comparable results for both agents.23-25
A recent addition to the armamentarium for intravitreal treatment of AMD is the FDA-approved aflibercept (Eylea). It is a recombinant fusion protein comprised of domains from human VEGF receptors 1 and 2. It binds to VEGF-A and VEGF-B, as well as placental growth factor. Findings from the initial clinical trials demonstrated significant efficacy for neovascular AMD.26
With durability of treatment and reduction of patient burden—especially due to the pandemic—a variety of treatment protocols have been examined.26 Depending on individual circumstances, many of these treatments using other novel proteins could be applied to patients with neovascular AMD.27 Among these are brolucizumab (Beovu), a humanized single-chain antibody fragment inhibitor of all isoforms of VEGF-A approved by the FDA in 2019, and faricimab (Vabysmo), the first bispecific antibody designed for intraocular use. A novel humanized bispecific IgG monoclonal antibody, faricimab independently binds to and neutralizes VEGF-A and angiopoietin-2 and was approved by the FDA in January 2022.28,29 Additional candidate molecules and delivery systems have been reviewed recently.27
Although there is considerable optimism over the treatments for neovascular AMD, a neglected area until recently has been non-neovascular AMD and GA. Several promising approaches are being investigated in this disorder of the retinal pigment epithelium.30 These approaches include elamipretide, an oral medication not approved in the United States but approved in the European Union, which targets mitochondrial support.31
In addition, specifically targeting the complement cascade, pegcetacoplan is a pegylated, highly selective peptide that binds C3, preventing its cleavage and inhibiting progression of GA.32 Other complement inhibiting molecules are also under investigation.33 Finally, photobiomodulation is being investigated for treatment of dry AMD, and preliminary results are promising.34
The discussion so far has centered on treatment for active lesions or to slow progression of diagnosed cases. What has not been addressed is prevention, which may increase the awareness of management options for dry AMD. allow us to stave off the Grim Reaper’s knock at the door of visual performance. Stay tuned.
References
1. Eye health statistics. American Academy of Ophthalmology. Accessed October 21, 2022. https://www.aao.org/newsroom/eye-health-statistics#_edn4
2. Diabetic retinopathy tables. National Eye Institute. Updated February 7, 2020. Accessed October 21, 2022. https://www.nei.nih.gov/learn-about-eye-health/resources-for-health-educators/eye-health-data-and-statistics/diabetic-retinopathy-data-and-statistics/diabetic-retinopathy-tables
3. Glaucoma tables. National Eye Institute. Updated February 7, 2020. Accessed October 21, 2022. https://www.nei.nih.gov/learn-about-eye-health/resources-for-health-educators/eye-health-data-and-statistics/glaucoma-data-and-statistics/glaucoma-tables
4. Prevalence of age-related macular degeneration (AMD). CDC. Updated October 31, 2022. Accessed December 28, 2022. https://www.cdc.gov/visionhealth/vehss/estimates/amd-prevalence.html
5. Eye health data and statistics. National Eye Institute. Updated June 15, 2022. Accessed December 28, 2022. https://nei.nih.gov/eyedata/amd
6. Prevent Blindness declares February as Age-related Macular Degeneration (AMD) and Low Vision Awareness Month. Prevent Blindness. January 26, 2022. Accessed December 28, 2022. https://preventblindness.org/February-age-related-macular-degeneration-amd-low-vision/
7. Cervantes-Castañeda RA, Banin E, Hemo I, Shpigel M, Averbukh E, Chowers I. Lack of benefit of early awareness to age-related macular degeneration. Eye (Lond). 2008;22(6):777-781. doi:10.1038/sj.eye.6702691
8. Ciulla TA, Hussain RM, Pollack JS, Williams DF. Visual acuity outcomes and anti-vascular endothelial growth factor therapy intensity in neovascular age-related macular degeneration patients: a real-world analysis of 49 485 eyes. Ophthalmol Retina. 2020;4(1):19-30. doi:10.1016/j.oret.2019.05.017
9. Heier JS, Brown DM, Shah SP, et al. Intravitreal aflibercept injection vs sham as prophylaxis against conversion to exudative age-related macular degeneration in high-risk eyes: a randomized clinical trial. JAMA Ophthalmol. 2021;139(5):542-547. doi:10.1001/jamaophthalmol.2021.0221
10. Neely DC, Bray KJ, Huisingh CE, Clark ME, McGwin G Jr, Owsley C. Prevalence of undiagnosed age-related macular degeneration in primary eye care. JAMA Ophthalmol. 2017;135(6):570-575. doi:10.1001/jamaophthalmol.2017.0830
11. Ferris FL 3rd, Wilkinson CP, Bird A, et al; Beckman Initiative for Macular Research Classification Committee. Clinical classification of age-related macular degeneration. Ophthalmology. 2013;120(4):844-851. doi:10.1016/j.ophtha.2012.10.036
12. Venhuizen FG, van Ginneken B, van Asten F, et al. Automated staging of age-related macular degeneration using optical coherence tomography. Invest Ophthalmol Vis Sci. 2017;58(4):2318-2328. doi:10.1167/iovs.16-20541
13. Brown GC. Vision and quality-of-life. Trans Am Ophthalmol Soc. 1999;97:473-511.
14. Recurrent choroidal neovascularization after argon laser photocoagulation for neovascular maculopathy. Macular Photocoagulation Study Group. Arch Ophthalmol. 1986;104(4):503-512. doi:10.1001/archopht.1986.01050160059012
15. Argon laser photocoagulation for neovascular maculopathy. Three-year results from randomized clinical trials. Macular Photocoagulation Study Group. Arch Ophthalmol. 1986;104(5):694-701. doi:10.1001/archopht.1986.01050170084028
16. Wormald R, Evans J, Smeeth L, Henshaw K. Photodynamic therapy for neovascular age-related macular degeneration. Cochrane Database Syst Rev. 2007;(3):CD002030. doi:10.1002/14651858.CD002030.pub3
17. Folkman J. Endogenous angiogenesis inhibitors. APMIS. 2004;112(7-8):496-507. doi:10.1111/j.1600-0463.2004.apm11207-0809.x
18. Judah Folkman, MD. eye.hms.harvard.edu. Accessed March 23, 2023. https://eye.hms.harvard.edu/judahfolkman
19. Gragoudas ES, Adamis AP, Cunningham ET Jr, Feinsod M, Guyer DR; VEGF Inhibition Study in Ocular Neovascularization Clinical Trial Group. Pegaptanib for neovascular age-related macular degeneration. N Engl J Med. 2004;351(27):2805-2816. doi:10.1056/NEJMoa042760
20. Cunningham ET Jr, Adamis AP, Altaweel M, et al; Macugen Diabetic Retinopathy Study Group. A phase II randomized double-masked trial of pegaptanib, an anti-vascular endothelial growth factor aptamer, for diabetic macular edema. Ophthalmology. 2005;112(10):1747-1757. doi:10.1016/j.ophtha.2005.06.007
21. Kourlas H, Schiller DS. Pegaptanib sodium for the treatment of neovascular age-related macular degeneration: a review. Clin Ther. 2006;28(1):36-44. doi:10.1016/j.clinthera.2006.01.009
22. Michels S, Schmidt-Erfurth U, Rosenfeld PJ. Promising new treatments for neovascular age-related macular degeneration. Expert Opin Investig Drugs. 2006;15(7):779-793. doi:10.1517/13543784.15.7.779
23. CATT Research Group; Martin DF, Maguire MG, Ying GS, Grunwald JE, Fine SL, Jaffe GJ. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med. 2011;364(20):1897-1908. doi:10.1056/NEJMoa1102673
24. Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group; Martin DF, Maguire MG, Fine SL, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012;119(7):1388-1398. doi:10.1016/j.ophtha.2012.03.053
25. Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group Writing Committee; Martin DF, Maguire MG, Fine SL, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2020;127(4S):S135-S145. doi:10.1016/j.ophtha.2020.01.029
26. Heier JS, Brown DM, Chong V, et al; VIEW 1 and VIEW 2 Study Groups. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology. 2012;119(12):2537-2548. Published correction appears in Ophthalmology. 2013;120(1):209-210.
27. Tan CS, Ngo WK, Chay IW, Ting DS, Sadda SR. Neovascular age-related macular degeneration (nAMD): a review of emerging treatment options. Clin Ophthalmol. 2022;16:917-933. doi:10.2147/OPTH.S231913
28. Mukai R, Matsumoto H, Akiyama H. Risk factors for emerging intraocular inflammation after intravitreal brolucizumab injection for age-related macular degeneration. PLoS One. 2021;16(12):e0259879. doi:10.1371/journal.pone.0259879
29. Regula JT, Lundh von Leithner P, Foxton R, et al. Targeting key angiogenic pathways with a bispecific CrossMAb optimized for neovascular eye diseases. EMBO Mol Med. 2016;8(11):1265-1288. Published correction appears in EMBO Mol Med. 2019;11(5):e10666.
30. Bird A. Role of retinal pigment epithelium in age-related macular disease: a systematic review. Br J Ophthalmol. 2021;105(11):1469-1474. doi:10.1136/bjophthalmol-2020-317447
31. Allingham MJ, Mettu PS, Cousins SW. Phase 1 clinical trial of elamipretide in intermediate age-related macular degeneration and high-risk drusen: ReCLAIM high-risk drusen study. Ophthalmol Sci. 2021;2(1):100095. doi:10.1016/j.xops.2021.100095
32. Liao DS, Grossi FV, El Mehdi D, et al. Complement C3 inhibitor pegcetacoplan for geographic atrophy secondary to age-related macular degeneration: a randomized phase 2 trial. Ophthalmology. 2020;127(2):186-195. doi:10.1016/j.ophtha.2019.07.011
33. Jaffe GJ, Westby K, Csaky KG, et al. C5 inhibitor avacincaptad pegol for geographic atrophy due to age-related macular degeneration: a randomized pivotal phase 2/3 trial. Ophthalmology. 2021;128(4):576-586. doi:10.1016/j.ophtha.2020.08.027
34. Markowitz SN, Devenyi RG, Munk MR, et al. A double-masked, randomized, sham-controlled, single-center study with photobiomodulation for the treatment of dry age-related macular degeneration. Retina. 2020;40(8):1471-1482. doi:10.1097/IAE.0000000000002632
2 Commerce Drive
Cranbury, NJ 08512
All rights reserved.