- February digital edition 2020
- Volume 12
- Issue 2
Your glaucoma patients also have ocular surface disease
Employ treatment strategies that control both conditions with less reliance on drops.
Reducing the medication burden on patients is important for compliance and overall quality of life, and helps to strengthen doctor–patient relationships. Use of the Prokera cryo-preserved amniotic membrane, in conjunction with new imaging technologies, can reduce dry eye symptoms for five to seven months.
It is a fact:
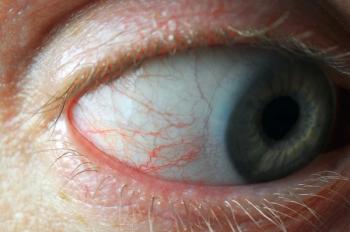
Glaucoma affects nearly 3 million Americans, including one in 50 people aged 40 years and older, according to the American Academy of Ophthalmology. Worldwide, an estimated 76 million people will have the disease in 2020. Many patients with glaucoma have signs and symptoms of ocular surface disease (OSD)-according to one study, about 56.9 percent of women and 45.7 percent of men.1
Considering that both glaucoma and OSD occur more often as patients age, and the most common form of treatment for glaucoma are drops presesrved with benzalkonium chloride (BAK), this information is not a surprise. ODs have grown accustomed to seeing glaucoma patients with their injected conjunctivae and keratitis.
When treating
Complex drug dosing regimens are a significant barrier to patients’ adherence to therapy.2,3 For glaucoma in general, patients’ lack of compliance ranges from 23 percent to as high as 60 percent.4-8
Compliance matters
Think about how this changes when it comes to OSD patients. Practitioners prescribe as if patients are compliance superheroes. We put these folks on artificial tears at least four times a day, plus prescription medications like cyclosporine (Restasis, Allergan; Cequa, Sun Pharma; Klarity-C, ImprimisRx) or lifitegrast (Xiidra, Novartis) twice a day, not to mention doxycycline for some.
We tell them to use heat masks, hot compresses, and other interventions. Then we believe patients will follow through despite all the data to the contrary.
At least OSD patients have the advantage that treatments are likely to help them feel and see better. Unfortunately, this can’t be said for glaucoma patients. No matter how well ODs manage the disease-
Innovative paradigm
Optometrists have a great opportunity to treat glaucoma patients’ OSD by offering therapy that provides results they can see and feel. We can get out of our prescribing rut with
We can do much to improve our patients’ OSD by altering their
Another way to reduce preservatives is to eliminate a drop-or drops-entirely. Selective laser trabeculoplasty (SLT) has been available for years but continues to be underutilized. Research suggests evidence for considering laser as first-line treatment, although it estimates that only 15 percent to 20 percent of physicians take this approach.9,10 Even the early Glaucoma Laser Trial (GLT) comparing the use of the old argon laser trabeculoplasty to medications as a first-line treatment found that patients starting with laser had better long-term outcomes than patients starting with medications.11 Patients were also using fewer drops at the end of follow-up.
New developments in treatment are shifting away from topical drops as first-line treatment, replacing them with delivery systems that include punctal plugs, rings, contact lenses, and injections. It should also be routine to consider minimally invasive glaucoma surgery (MIGS) for any glaucoma patient having cataract surgery. The iStent (Glaukos) is FDA-approved only for implantation during cataract surgery; other MIGS devices are available. Xen gel stent (Allergan) and endocyclophotocoagulation (ECP) are options; although all of these are beyond the scope of this article.
Innovative methods
Of course, some
These treatments put a heavy burden on patients, especially those who are also being treated for glaucoma. We can expect the compliance on both fronts to suffer, with a negative impact on the efficacy of treatment for both conditions, not to mention the patients’ quality of life.
A fresh approach
With the development of InflammaDry (Quidel) testing for the inflammatory marker MMP-9, TearLab’s tear osmolarity testing, and the LipiScan Dynamic Meibomian Imager (Johnson & Johnson Vision), we can obtain more detailed information regarding patients’ corneal health.
Using these new diagnostic tools combined with our standbys of tear break-up time (TBUT), fluorescein and lisaamine green staining, meibomian gland expression, and a good clinical examination, we can drill down to the root cause of OSD. Gone are the days of every dry eye patient starting on artificial tears. Instead, practitioners tailor treatments to address the components of the disease based on clinical information.
Hitting the market along with these advanced diagnostics are the additional topical therapies lifitegrast (Xiidra, Novartis) and new higher-concentration cyclosporine 0.09% (Cequa, Sun Pharma). Plus, practitiones can opt for procedures to treat dry eye like LipiFlow (Johnson & Johnson Vision), BlephEx (RySurg), intense pulsed light (IPL), and iLux (Alcon).
Drop-free treatment
I developed my Drop-Free
DDET recognizes that it is essential to accurately assess the cause of OSD to provide the correct targeted treatment. InflammaDry indicates the presence of the inflammatory marker MMP-9; therefore, if the clinical examination agrees, we can avoid unnecessary anti-inflammatory topical medications. Punctal occlusion can be considered for those with non-inflammatory disease because it is effective for reducing symptoms of DED and reduces compliance concerns.14
LipiScan meibography combined with meibomian gland expression and TBUT help when assessing OSD mechanisms. Meibomian gland dysfunction (MGD) has been reported to be present in 86 percent of patients with OSD.15 In my experience, identifying and treating MGD is an effective way to win patients and develop strong doctor–patient relationships. MGD treatment with LipiFlow, intense pulsed light (IPL), or iLux can yield a stabilized tear film, increased TBUT, enhanced comfort, and improved vision. A trained technician can perform these procedures while the doctor continues to see patients. Minimal at-home maintenance using a heat mask and lid hygiene extend the effects of treatment.
Amniotic membrane
I educate my OSD patients about the cause and effect of their dry eyes. MGD is often the cause, and the effect is frequently seen as superficial punctate keratitis. Their unhealthy corneal epithelium results in reduced visual acuity and discomfort. Many patients with chronic DED have severe keratitis but no discomfort due to their corneas becoming neurotrophic.16
Incorporating amniotic membrane treatment with CAM has changed the way I treat OSD. I had long used it to treat standard corneal injuries like burns and abrasions, with corneas healing in a matter of days. I opted to move CAM treatment from the end of the line for patients with severe DED up to a first-line treatment. My OSD patients are very receptive to treating their
Prokera also has anti-inflammatory properties17,18 and, as such, it has replaced the use of topical anti-inflammatory drops such as Restasis and Xiidra for most of my patients. The occasional placement of the membrane reduces inflammation as measured by InflammaDry and clinical observation.
In practice, I have patients wear Prokera Clear for five days, then treat the contralateral eye for another five days. I use the monocular approach so patients can proceed with their normal routine. The patient’s vision is only moderately affected if the membrane aperture doesn’t line up with their line of sight. If keratitis is still present after removing the CAM at the end of five days, the patient and I decide whether to treat for another two to five days immediately or move to the second eye and return to the first eye later. I have found that many patients have a reduction in symptoms for five to seven months following treatment. When symptoms return, I typically treat with a shorter course of a two-day placement, again monocularly and treating the other eye successively. Patients are happy to get their ‘booster’ treatment and have their symptoms resolve, remembering the success of the previous treatment.
For patients with severe glaucoma who need multiple drops to be controlled or those resistant to a change in treatment that has worked for years, I have found Prokera can be a useful way to treat BAK-induced keratitis. Occasional CAM placement throughout the year can allow the effective glaucoma treatment to remain unchanged. I choose Prokera Clear for glaucoma patients in order to not occlude their vision and to have a clear opening for topical
Much like the move to making SLT a first-line treatment, by using CAM at the beginning of dry eye treatment we do our patients a tremendous service and build our practices with devoted patients who have healthier eyes.
Fewer drops, more success
As doctors, we have a great impact on our patients’ quality of life. We can tie them down with medications requiring frequent treatments, copays, and trips to the pharmacy or we can embrace advances and treat them accordingly. By getting out of our prescribing rut and writing prescriptions for BAK-free medications, we can reduce our patients’ chances of developing OSD. By performing SLT earlier, we can avoid or reduce self-administered medications and their associated compliance challenges. If we adopt a procedure-based approach to
References:
1. Pflugfelder SC, Baudouin C. Challenges in the clinical measurement of ocular surface disease in glaucoma patients. Clin Ophthalmol. 2011;5:1575–1583. doi: 10.2147/OPTH.S24410
2. Tsai JC. A comprehensive perspective on patient adherence to topical glaucoma therapy. Ophthalmology 2009;116:S30–S36.
3. Nordstrom BL, Friedman DS, Mozaffari E, Quigley HA, Walker AM. Persistence and adherence with topical glaucoma therapy. Am J Ophthalmol 2005;140:598–606.
4. Richardson C, Brunton L, Olleveant N, et al. A study to assess the feasibility of undertaking a randomized controlled trial of adherence with eye drops in glaucoma patients. Patient Prefer Adherence 2013;7:1025–1039.
5. Gooch N, Molokia SA, Condie R, et al. Ocular drug delivery for glaucoma management. Pharmaceutics 2012;4:197-211.
6. Mansberger SL, Sheppler CR, McClure TM, et al. Psychometrics of a new questionnaire to assess glaucoma adherence: The glaucoma treatment compliance assessment tool. Trans Am Ophthalmol Soc. 2013;111:1-16.
7. Budenz DL. A clinician’s guide to the assessment and management of nonadherence in glaucoma. Ophthalmology. 2009;116:S43–S47.
8. Sleath B, Blalock S, Covert D, et al. The relationship between glaucoma medication adherence, eye drop technique, and visual field defect severity. Ophthalmology. 2011;118:2398–2402.
9. Li X, Wang W, Zhang X. Meta-analysis of selective laser trabeculoplasty versus topical medication in the treatment of open-angle glaucoma. BMC Ophthalmol. 2015;15:107.
10. Wong MO, Lee JW, Choy BN, et al. Systematic review and meta-analysis on the efficacy of selective laser trabeculoplasty in open-angle glaucoma. Surv Ophthalmol. 2015;60:36–50.
11. The Glaucoma Laser Trial (GLT) and glaucoma laser trial follow-up study: 7. Results. Glaucoma Laser Trial Research Group. [No authors listed] Am J Ophthalmol. 1995;120718–731.
12. The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5:75-92.
13. Nelson JD, Craig JP, Akpek EK et al. TFOS DEWS II Introduction. Ocul Surf. 2017;15:269-275.
14. Jones L, Downie LE, Korb D, et al. TFOS DEWS II management and therapy report. Ocul Surf. 2017 Jul;15(3):575-628.
15. Lemp MA, Crews LA, Bron AJ, Foulks GN, Sullivan BD. Distribution of aqueous-deficient and evaporative dry eye in a clinic-based patient cohort: a retrospective study. Cornea. 2012; 31 (5): 472-478.
16. Goyal S, Hamrah P. Understanding Neuropathic Corneal Pain-Gaps and Current Therapeutic Approaches. Semin Ophthalmol 2016;31:59–70.
doi: 10.3109/08820538.2015.1114853
17. McDonald MB, Sheha H, Tighe S, et al. Treatment outcomes in the DRy Eye Amniotic Membrane (DREAM) study. Clin Ophthalmol 2018;12:677–681. doi: 10.2147/OPTH.S162203.
18. Tseng SCG. HC-HA/PTX3 purified from amniotic membrane as novel regenerative matrix: insight into relationship between inflammation and regeneration. Invest Ophthalmol Vis Sci 2016;57:ORSFh1–ORSFh8. doi: 10.1167/iovs.15-17637.
Articles in this issue
almost 6 years ago
How ODs should handle non-compliance with contact lens replacementalmost 6 years ago
Prevention, artificial intelligence, and the future of optometryalmost 6 years ago
Laser vision correction: Look backward to move forwardalmost 6 years ago
Glaucoma facts: Essential perspectives for long-term managementalmost 6 years ago
Technological advancements in glaucoma managementalmost 6 years ago
Experts offer advice to ODs starting dry eye subspecialtyalmost 6 years ago
ODs must raise awareness about glaucomaalmost 6 years ago
Retinal detachment seals itselfalmost 6 years ago
The myopia epidemic: Are new therapies in sight?almost 6 years ago
A forecast of the global ocular allergy marketNewsletter
Want more insights like this? Subscribe to Optometry Times and get clinical pearls and practice tips delivered straight to your inbox.