- September digital edition 2021
- Volume 13
- Issue 9
5 myths and misunderstandings about cross-linking and keratoconus
Know the facts, not misinformation, about slowing or halting disease progression
Corneal collagen cross-linking has been available in the United States for several years, with an excellent record of slowing or halting progression of keratoconus and postrefractive ectasia. However, misunderstandings remain. Let’s address several myths about the disease and the procedure.
Myth #1: I DON’T HAVE THE RIGHT EQUIPMENT TO DIAGNOSE KERATOCONUS
In an ideal world, everyone managing keratoconus would have topography and tomography available to them. However, even clinicians who don’t have topography and/or tomography in the office can still watch for warning signals in 4 key areas:
» Genetic or systemic disease risk factors. In the CLEK study (NCT00000155), 14% of those with keratoconus had a positive family history for the disease.1 Down syndrome and connective tissue diseases, such as rheumatoid arthritis, Marfan syndrome, and lupus, also predispose a patient to keratoconus. A noninvasive cheek swab test (AvaGen; Avellino) is commercially available to optometrists interested in a genetic test that examines over 1000 variants across 75 genes for keratoconus.
» Environmental risk factors. Half of the patients with keratoconus in the CLEK study reported vigorous eye rubbing, and 53% had a history of atopy.1
» New symptoms in an established patient. This could include decreased tolerance to contact lenses or new difficulty seeing at night because of ghosting or halos around lights.
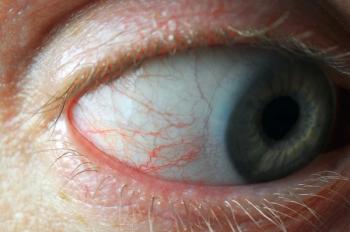
» Exam findings. At the slit lamp, any evidence of central corneal thinning, such as a thinning of the optic section as you sweep across the cornea, Fleischer ring, corneal scarring, or Vogt lines/vertical striae (Figure) should be closely evaluated. New, or newly irregular, astigmatism and poor or worsening best-corrected visual acuity for which there is no other explanation are also concerning. If you perform retinoscopy, the oil droplet reflex (Charleux sign) is a red flag.
The presence of any of these risk factors or signs should warrant a referral to an optometrist who has an advanced topographer/tomographer or to a practice that is performing cross-linking. Glaukos offers a program called iDetect KC that allows optometrists to use an advanced topographer to diagnose keratoconus and participate in a keratoconus data registry.
Myth #2: PATIENTS WHO CAN BE MANAGED WITH CONTACT LENSES DON’T NEED CROSS-LINKING
Historically, ODs managed the vision of patients with keratoconus with spectacles or soft contact lenses, moved them into specialty contact lenses when more traditional methods failed, then finally resorted to a referral for full-thickness penetrating keratoplasty (PKP). When PKP was the only surgical answer, it made sense to wait to refer. A PKP is invasive and may need to be repeated more than once in a patient’s life.
Results from a 2011 study reported survival rates of PKP for keratoconus as follows: At 20 years after surgery, 51% of transplants were failing, and at 23 years, 83% of transplants were failing.2 With FDA-approved corneal cross-linking, which has the potential to slow or halt the progression of the disease, it no longer makes sense to watch and wait. Contact lenses—even flat-fitting gas permeable lenses—do not stop progression of keratoconus.3 Patients with progressive keratoconus are best served by cross-linking as soon as possible to preserve their vision.
Conversely, it is also false that patients who have undergone cross-linking won’t need spectacles, soft contact lenses, or specialty contact lenses. Patients with keratoconus in the pivotal US cross-linking clinical trial achieved an average 1.60 D of corneal flattening,4 so there is some effect on vision, but the procedure is designed to slow or halt progression, not to correct vision. Optometrists should expect that patients who wore specialty contact lenses before cross-linking will still need them afterward, although the prescription may need to be adjusted.
Myth #3: CROSS-LINKING IS AN OUT-OF-POCKET PROCEDURE
As with many new technologies, cross-linking was not widely covered by medical insurance when it was first introduced, but that has changed. Today, most of commercial health plans in the United States, representing 97% of commercially covered lives, consider cross-linking with FDA-approved technology a covered service for progressive keratoconus. Currently the only FDA-approved drug/device combination is the iLink platform (Glaukos), which includes the KXL System, Photrexa, and Photrexa Viscous.
As with any technology or treatment, it is important to understand the history and data behind each. The pivotal US cross-linking clinical trial provided data showing that epithelium (epi)-off cross-linking slows or halts progression and, on average, achieves corneal flattening.4 Once ODs have seen and believe in the data, it is important to advocate for patients in a way that allows the cross-linking procedure to be covered under their commercial insurance plan as a covered medical procedure.
The key to successful reimbursement is document progression. Although this varies by region and carrier, one typically must satisfy 1 or more of these 5 criteria:
• At least 1.00-D increase in the steepest keratometry value
• At least 1.00-D increase in regular astigmatism
• At least 0.50-D myopic shift in the subjective manifest refraction
• At least 0.1-mm decrease in the back optical zone radius for a rigid gas permeable lens, where other information is not available
• Failure of conservative treatment (spectacles, gas permeable lenses) to adequately correct vision
Myth #4: EPI-ON IS THE PREFERRED PROCEDURE
Although some attractive potential benefits of transepithelial or epi-on cross-linking exist, this procedure is not FDA approved in the United States. In theory, the potential advantages of an epi-on procedure include a shorter procedure and faster or more comfortable healing. Unfortunately, the epithelium is extraordinarily effective at doing its job of keeping things out of the eye. An intact epithelium is a barrier to all 3 of the necessary components in cross-linking—riboflavin, UV rays, and oxygen—reaching the stroma. Because of this, most research has shown that superior results are achieved with the epi-off or Dresden protocol that is approved in the US.5-7
Numerous strategies to overcome these challenges are under investigation, including accelerated and pulsed UV light delivery, specialized goggles to deliver supplemental oxygen to the cornea, and iontophoresis or the addition of components that increase the penetration of riboflavin solutions. If these innovations can overcome the challenges of an intact epithelium, it is possible that epi-on may one day supplant epi-off as the preferred procedure, but that hasn’t happened yet.
Recently announced were topline results from a US phase 3 epi-on cross-linking study, in which the primary end point of a 1.00-D effect on Kmax in the treatment group compared with placebo was met.8 Although the industry waits for additional results of clinical trials incorporating these investigational approaches, optometrists should feel comfortable with the proven safety and efficacy of epi-off cross-linking and recommending it to their patients.3-6
Myth #5: IF I REFER A PATIENT FOR CROSS-LINKING, THEY WON’T COME BACK
Collaborative care of patients with keratoconus is a win-win-win. The ophthalmologist can perform more surgery without spending a lot of time on postoperative care. The optometrist builds a relationship of trust with the patient by identifying keratoconus, referring for treatment, and continuing to manage that patient over time. The patient certainly benefits from slowing or halting progression of a sight-threatening disease.
After cross-linking, patients with keratoconus require ongoing monitoring. Although most patients remain stable, a recent analysis has shown stability up to a decade after treatment.9 Progression may be more likely among those who present with the disease as children or in their younger years10-12 or who had very steep corneas (Kmax ≥ 58 D) prior to cross-linking.13 Even with great stability, patients with keratoconus still need vision correction, often with specialty lenses.
The key to comanaging cross-linking effectively is good communication with the surgeon to let them know you are comfortable and want to continue to manage the patient post cross-linking. Incidentally, postoperative appointments can be billed to medical insurance as office visits because no global period exists for cross-linking.
Conclusion
Keratoconus care has considerably evolved in recent years. This is an exciting time for patients, who stand to benefit greatly from cross-linking, and for optometrists as they navigate the changing protocols for cross-linking and keratoconus.
References
1. Wagner H, Barr JT, Zadnik K. Collaborative longitudinal evaluation of keratoconus (CLEK) study: methods and findings to date. Cont Lens Anterior Eye. 2007;30(4):223-232. doi:10.1016/j.clae.2007.03.001
2. Kelly TL, Williams KA, Coster DJ; Australian Corneal Graft Registry. Corneal transplantation for keratoconus: a registry study. Arch Ophthalmol. 2011;129(6):691-697. doi:10.1001/archophthalmol.2011.7
3. Downie LE, Lindsay RG. Contact lens management of keratoconus. Clin Exp Optom. 2015;98(4):299-311. doi:10.1111/cxo.12300
4. Hersh PS, Stulting RD, Muller D, Durrie DS, Rajpal RK; US Crosslinking Study Group. United States multicenter clinical trial of corneal collagen crosslinking for keratoconus treatment. Ophthalmology. 2017;124(9):1259-1270. doi:10.1016/j.ophtha.2017.03.052
5. Kobashi H, Rong SS, Ciolino JB. Transepithelial versus epithelium-off corneal crosslinking for corneal ectasia. J Cataract Refract Surg. 2018;44:12:1507-1516. doi:10.1016/j.jcrs.2018.08.021
6. Choi M, Kim J, Kim EK, Seo KY, Kim TI. Comparison of the conventional Dresden protocol and accelerated protocol with higher ultraviolet intensity in corneal collagen cross-linking for keratoconus. Cornea. 2017;36(5):523-529. doi:10.1097/ICO.0000000000001165
7. Rush SW, Rush RB. Epithelium-off versus transepithelial corneal collagen crosslinking for progressive corneal ectasia: a randomised and controlled trial. Br J Ophthalmol. 2017;101(4):503-508. doi:10.1136/ bjophthalmol-2016-308914
8. Glaukos announces positive phase 3 trial results for iLink epi-on investigational therapy. Press release. Eyewire. February 25, 2021. Accessed August 10, 2021. https://eyewire.news/articles/ glaukos-announces-positive-phase-3-trial-results-for-ilink-epi-on-investigational-therapy/
9. Gelles JD, Huang S, Greenstein SA, Hersh PS. Corneal collagen crosslinking: 10 year follow up for patients with keratoconus and post refractive surgery corneal ectasia. Poster presented at: American Academy of Optometry Annual Meeting; October 7-22, 2020. Accessed August 10, 2021. https://www.aaopt.org/detail/knowledge-base-article/ corneal-collagen-crosslinking-10-year-follow-up-for-patients-with-keratoconus-and-post-refractive-surgery-corneal-ectasia
10. Buzzonetti L, Bohringer D, Liskova P, Lang S, Valente P. Kertatoconus in children: a literature review. Cornea. 2020;39(12):1592-1598. doi:10.1097/ICO.0000000000002420
11. Barbisan PRT, Pinto RDP, Gusmão CC, de Castro RS, Arieta CEL. Corneal collagen cross-linking in young patients for progressive keratoconus. Cornea. 2020;39(2):186-191. doi:10.1097/ ICO.0000000000002130
12. Shetty G, Behshad S. Corneal crosslinking outcomes in children with progressive keratoconus. Paper presented at: American Society of Cataract and Refractive Surgeons Annual Meeting; May 16-17, 2020; Virtual.
13. Kuechler SJ, Tappeiner C, Epstein D, Frueh BE. Keratoconus progression after corneal cross-linking in eyes with preoperative maximum keratometry values of 58 diopters and steeper. Cornea. 2018;37(11):1444-1448. doi:10.1097/ICO.0000000000001736
Articles in this issue
over 4 years ago
CASE REPORT: Self-induced maculopathy by 12-year-old boyover 4 years ago
ODs are ignoring macular degenerationover 4 years ago
Angle closure presentation differsover 4 years ago
Pupil size matters in presbyopia treatmentover 4 years ago
Aesthetics and eye care are a natural fitNewsletter
Want more insights like this? Subscribe to Optometry Times and get clinical pearls and practice tips delivered straight to your inbox.















































