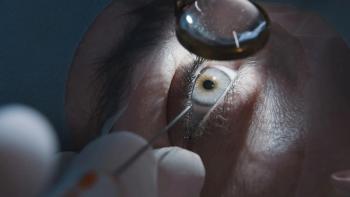
Nicox launches Sjögren’s diagnostic test
Sjö is a proprietary laboratory test developed by Immco Diagnostics Inc. which combines traditional markers with three novel, proprietary biomarkers, allowing earlier detection of the disease.
Sophia Antipolis, France-
Sjö is a proprietary laboratory test developed by Immco Diagnostics Inc. which combines traditional markers with three novel, proprietary biomarkers, allowing earlier detection of the disease.
Nicox has partnered with the Sjögren's Syndrome Foundation to raise awareness of the prevalence, seriousness, and significance of Sjögren's Syndrome to eyecare professionals. A targeted media campaign over the next few months will aim to educate about the disease and the role they can play in early detection.
Newsletter
Want more insights like this? Subscribe to Optometry Times and get clinical pearls and practice tips delivered straight to your inbox.