Norlase Leaf laser receives CE mark approval
Distribution to begin in the European Union.
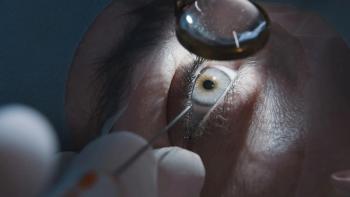
Norlase has been granted the European CE mark for its Leaf laser, following a 510(l)-market clearance by the US Food and Drug Administration (FDA) and ISO 13485:2016 certification. This approval will allow the company to market and distribute its fiberglass, green laser for the treatment of retina and glaucoma diseases in the European Union and other countries that recognize the European standard.
“This marks another significant milestone as we continue our mission to improve practice efficiency and workflow for ophthalmologists and equip them with the tools to provide quality eye care for patients worldwide,” said Oliver Hvidt, Norlase president, in a statement.
The Leaf laser photocoagulator allows ophthalmologists to perform laser therapy with minimal set-up time and physical space because it attaches to an existing slit lamp. It connects to a wireless tablet as its user interface and offers voice control features. Physicians can operate it touch-free.
Newsletter
Want more insights like this? Subscribe to Optometry Times and get clinical pearls and practice tips delivered straight to your inbox.