Retina
Latest News

Latest Videos

CME Content
More News

Two recent journal articles underscore the organ’s importance in identifying systemic disease processes.

Understanding AMD pathophysiology through nutrition has helped shape the disease’s detection and management.

Investigators presented genomic, transcriptomic, histological and functional evidence that the Greenland shark retains an intact visual system well-adapted for life in dim light.

Utilize oral agents, diet, and glucose monitoring systems when acting to preserve vision.

Knowing what’s on the market for AMD and GA aids in preserving vision.

The FDA issued a CRL for ONS-5010, citing the need for additional confirmatory efficacy evidence despite previous trials showing efficacy, according to the company.

A recent study represents a step forward in investigations of the retinal layers as they are affected by glaucoma.
RGCs can offer a window into confirming eye condition diagnoses.
Outlining the benefits of modulating the gut microbiome in diabetes.

Additionally, the FDA approved a monthly dosing option for some patients who may benefit from resuming this schedule across approved indications.

Maturing of the vestibulo-ocular reflex is independent of sensory input.

Raman Bhakhri, OD, FAAO; Julie Rodman, OD, MS, FAAO; and Andrew Rixon, OD, provide retinal updates during the meeting.

Using storytelling to highlight inherited eye disease awareness: Inside the film "My Eyes"
The optometrist–turned filmmaker, Tsu Shan Chambers, discusses how her feature aims to improve public understanding of inherited retinal disease.

Eye care providers are uniquely positioned to catch early risks during routine exams.

Rodman is part of the faculty for a CE Spotlight Symposium that will focus on how extending dosing intervals can enhance the quality of life for patients with neovascular retinal disease.

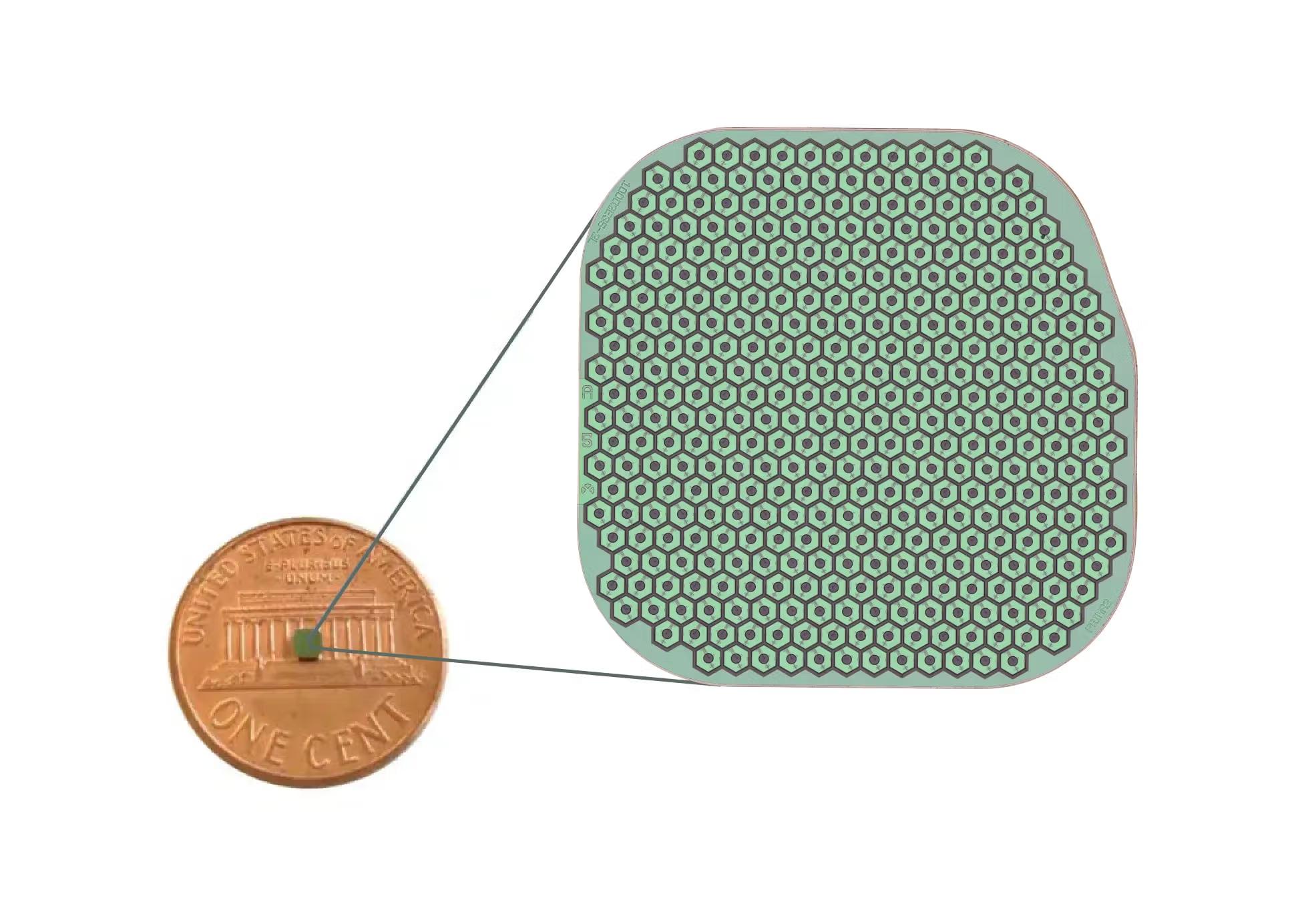
Researchers will investigate a gene in the eye that is crucial for normal vision, but can cause retinal diseases when mutated that often lead to blindness.

FDA issues a complete response letter to Outlook Therapeutics for ONS-5010, citing insufficient evidence of effectiveness for wet AMD treatment.

Researchers found that patients with primary open-angle glaucoma have less agmatine and thiamine in the aqueous humor than patients without glaucoma.

Researchers used adaptive optics scanning light ophthalmoscopy (AOSLO) to study retinas in mice with photoreceptor damage, specifically photoreceptor laser injury.

A rare case of tubulointerstitial nephritis and uveitis syndrome in a teenager highlights the importance of multidisciplinary diagnosis and management.
Ocular salvage and preservation of vision are essentially dependent on early diagnosis, and several studies have been conducted to identify the factors leading to delayed diagnosis at presentation.

Better best-corrected visual acuity values and baseline thicker ganglion cell layer and retinal nerve fiber layer at ocular coherence tomography measurements were found to be the key predictive factors.

The latest in drug and technology developments and updates from July 2025.
The first-in-class, twice-daily topical formulation is intended for the treatment of nonproliferative diabetic retinopathy and diabetic macular edema.