Retina
Latest News
Latest Videos

CME Content
More News

CVD could impact physicians' ability to identify choroidal melanoma from choroidal nevus.
Mark Dunbar, OD, FAAO, emphasizes the critical role optometrists play in the collaborative care of diabetic eye disease.


Dr. Ferrucci summarizes his Vision Expo West presentations and shares clinical pearls for managing retinal cases.

From schools for blind individuals to early intervention services, families have options to help prepare their children with visual impairments for adulthood.
By identifying progressive GA earlier, primary eye care providers can make timely referrals and save patients’ visual function.

Living a long life and preserving eye health can go hand in hand.

Researchers at the Medical College of Georgia at Augusta University are teaming up with Polaris Dawn to investigate how eye changes astronauts may experience during spaceflight could lead to multiple symptoms once back on Earth.

The product candidate is a modifier gene therapy for broad retinitis pigmentosa indication.
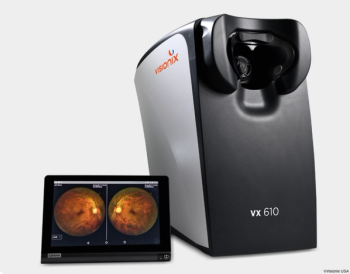
The device features cross-polarized light technology, allowing for image clarity without the need for pupil dilation.

Understanding the latest innovative technologies.

A research team for the $3.3 million study will assess the front and the back of the eye and different types of glucose dysfunction.

Some cases require unorthodox approaches.

It’s time to act with a few rational and relatively modest proposals.

Comanagement is a key component for treatment of disease.

The new smart glasses utilize artificial intelligence to visually assist those with age-related macular degeneration and other retinal disorders causing vision loss.

Case complexity underscores the importance of interdisciplinary communication.

The study helped identify predisposing factors for severe SANS in astronauts.

A new analysis by researchers at the National Institutes of Health shows the benefit of taking AREDS2 formula in late AMD.

The 2-year Phase III data demonstrates Susvimo’s potential as an alternative to eye injections to treat diabetic macular edema and diabetic retinopathy.

Study investigators conducted a case-control study to investigate the corneal topography parameters and biomechanics in patients with alopecia areata.
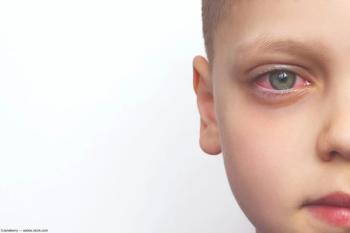
Pediatric TINU syndrome is rare, but responds well to treatment.
According to Genentech, faricimab-svoa is the first and only syringe that is prefilled with an FDA-approved bispecific antibody to treat wet AMD, DME and RVO.

The company also submitted a marketing authorization application with the European Medicines Agency.
Respecting the clinical importance of the pachychoroid spectrum is vital when analyzing OCT scans.